Validation of gene knockout/knockin in fly models by PCR screening with EzMate 401 Automated Pipetting System
Application note from WellGenetics Inc., Taiwan, by Dr. Yawen Chen
Introduction
CRISPR/Cas9 is a gene editing tool that can manipulate gene expression in plants, humans and animals. Compared to other existing genome editing methods, it is also faster, cheaper, more accurate, and more efficient. The CRISPR/Cas9 system is widely utilized in multiple areas of gene study at present.
In the workflow of CRISPR/Cas9 system, PCR screening is usually recommended to validate whether the gene editing is successful or not.
PCR preparation includes a complicated and subtle pipetting process. It always takes a lot of time and human error also occurs frequently. Using EzMate 401 automated pipetting system with CoolBlock adapters can not only save more manpower and time, but also improve the accuracy and precision by avoiding the human errors.
Materials
-
Consumable and Reagents
- 50 uL Robotic tips
- 8-strip tube
- 1.5 c.c. eppendorf
-
Equipment
- EzMate 401 with 1 channel Automated Pipetting Module (APM)
- CoolBlock adapters

Figure 1. EzMate 401 in the lab of WellGenetics Inc.
Methods
- Place different pairs of primers with 1.5 c.c. eppendorf in area A-A1, A-A2 to A-E1, A-E2 and plasmid DNA in area A-A3 and PCR master mix in
area A-A4 all on pre-cooled 20-well CoolBlock™ adapters. - Place empty 8-strip tubes in area B-A to E line and others with the DNAs of CRISPR fly models in area B-F line on pre-cooled 96-well CoolBlock™ adapters.
- Place 50 uL Robotic tips in area C & D.
- First, aspirate 16.6 uL PCR master mix from area A-A4 and dispense into the empty 8-strip tubes in area B on pre-cooled 96-well CoolBlock™ adapters by 1-channel APM.
- Aspirate 1.2 uL different pairs of primers from area A-A1, A-A2 to A-E1, A-E2 and dispense into the 8-strip tubes in area B-A to E line with PCR master mix.
- Aspirate 1 uL DNAs of CRISPR fly models in area B-F line and dispense into the 8-strip tubes in area B-A to E line.
- Aspirate 1 uL plasmid DNA from area A-A3 and dispense into 8-strip tubes in area B-C12 and B-D12 for control.
- At last, transfer the 8-strip tubes from area B to the PCR instrument.
_%20The%20worktable%20of%20Arise%20EzMate%20401.jpg)
Figure 2 (Left). The worktable of EzMate 401
_%20The%20interface%20of%20software%20of%20Arise%20EzMate%20401.jpg)
Figure 3 (Right). The interface of software of EzMate 401
Result
The result of the PCR is confirmed by gel electrophoresis in Figure 4. We can see the significant bands in the positive control and no bands in the negative control in the control group to prove that the result is credible. Moreover, in the experimental group, we can also easily determine which sample has the positive result. This is ideal gel electrophoresis data by using EzMate 401 automated pipetting system with CoolBlock adapters which is a great improvement on the accuracy of PCR experiments.
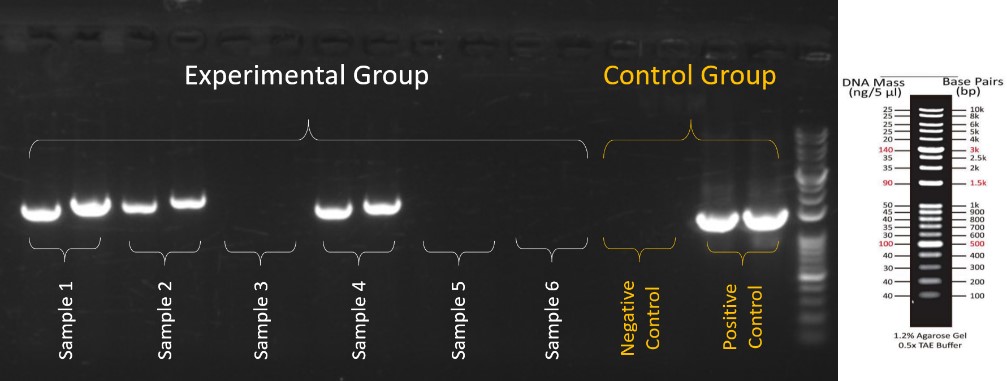
Figure 4. The PCR result in gel electrophoresis by EzMate 401
Conclusion
EzMate automated pipetting system is the best solution to handle the complicated and repeated pipetting process in the experiments of PCR preparation. It not only provides great reproducibility and repeatability of the experiment, but also easily solves the short-handed problem in lab. This system certainly makes time and manpower more efficient and also enhances the quality of eperimental data.