
Table of contents
- Advanced analysis of the application of adipose-derived stem cells in gelatin-based tissue engineering
Table of contents
- Advanced analysis of the application of adipose-derived stem cells in gelatin-based tissue engineering
Source: TSENG HSIANG LIFE SCIENCE LTD.
Cell migration generally refers to the process of cells moving from one place to another, which reflects the cellular condition in a certain environment. When the migration occurs, cells may develop into new tissues subsequently. In particular, significant cell migration may be induced by trauma or inflammation, which is a protective mechanism for wound repair and infection prevention. When the skin surface is broken, the resulting wound triggers the signaling pathways to stimulate the proliferation and migration of the peripheral fibroblasts towards the wound. Besides, inflammation arising from an infected wound releases special signals to attract the inflow of immune cells and further leads to phagocytosis and dissolution of the pathogens. As it should be, other than normal cell migration, cancer cells may also move from the in-situ organ to other locations, giving rise to metastasis.
Wound healing assay is one of the classic tests to observe cell migration. Traditionally, wound healing assay uses cultured cells with 90% confluency and applies a hard object like a tip on the medium to directly carve out several areas containing no cells for microscopically observing the cellular growth at such edges. However, manual scratching is difficult to maintain the sizes of the scratching areas and would cause certain harm to the cells. Therefore, culture inserts with fixed size were developed, which are intended to be removed after the cells have grown inside of it to produce fixed areas without cells for observation of the cellular growth.
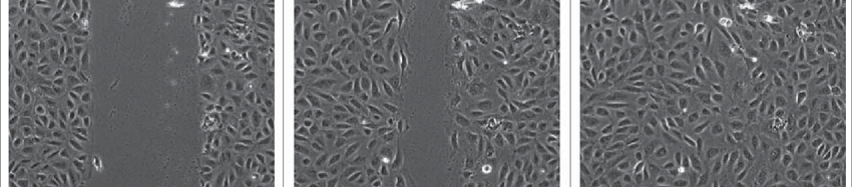
Figure 1. Typical images of wound healing assay
Nonetheless, either method requires repeatedly transferring the cells from the incubator for microscopic observation and recording. Yet frequent transferring makes specimens subject to continual environmental changes, and may instead produce experimental bias due to cellular stress responses. Furthermore, recurrent opening and closing of the incubator leads to environmental instability and increases the risk of contamination, which not only affect our experiment, but also the other ongoing studies in the lab.
Hence, the current observation frequency of wound healing assay is mainly once every 8-12 hours, which lowers the impact of environmental changes on the cells dramatically. Although the growth rate of cells is relatively slow, such a long observation interval may still give rise to another problem, i.e. the difficulty in capturing the critical moment. It is often noticed that the previous observation documents exuberant cell growth, whereas the next observation reveals a completely different situation, without any records during this period. Another disadvantage of prolonged observation intervals is the inability to set a fixed location for the image record. In fact, the imaged area of each observation differs, and the absence of “continuity” inevitably creates a doubtful space.
In order to satisfy the contradictory demands of simultaneously achieving “instantaneous” and “long-lasting” effects during wound healing assay, Blue-ray Biotech has developed the new generation of the live-cell imaging system. Designed specifically for the high temperature and high humidity environment within the incubator, the size of the microscopic imaging system has been reduced substantially to allow direct placement inside the incubator. Real-time cell monitoring can be accomplished via connection to the external computer, eliminating the need to open the incubator repetitively. In addition, the observation interval may be set based on individual requirements, offering full-range monitoring without missing any critical moment. One computer can control up to 4 live cell imaging system. The software is compatible with Windows 10 systems and various remote desktop applications to provide information about prompt cellular conditions and experimental status (by remote monitoring of the cell status).
In response to the advancement of regenerative medicine, wound healing assay may be considered as an important test to observe the effects of experimental treatments on cell migration. Nowadays, with diversified cellular experiments, what we need is not only the final results, but also the alterations occurred during the entire process. Thus, a continuous live-cell imaging system will certainly be the best answer to meet the current trend.
Except for wound healing assay, a continuous live-cell imaging system is more applicable for advanced analysis of tissue engineering. The following section includes testimonials from EzScope users:
Advanced analysis of the application of adipose-derived stem cells in gelatin-based tissue engineering
- Purpose of the experiment
The major research interest of this laboratory is cartilage tissue engineering, which uses biomaterials to prepare 3D bioscaffolds for the evaluation of the feasibility of tissue regeneration. The change in the cellular condition is commonly seen due to the microenvironment formed by different biomaterials. Therefore, it is necessary to perform precursor experiments and preliminary assessments to prepare for the 3D bioscaffold.
- Brief description of the process
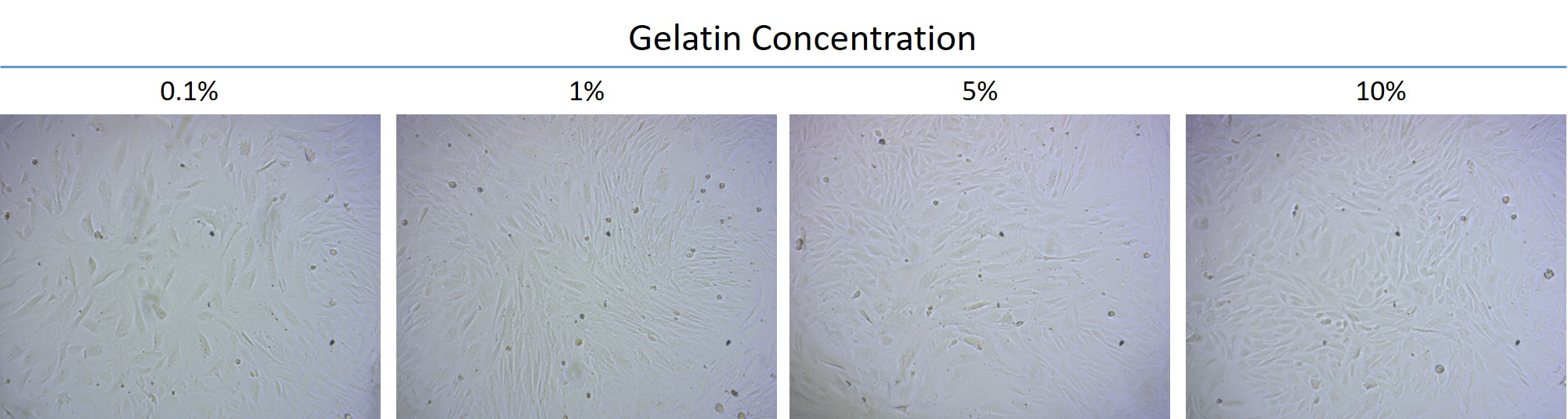
As the main research material, gelatin is coated on the culture plate with different concentrations ranging from 0.1% to 10% for subsequent observation. The cultured cells are the adipose-derived stem cells isolated from the subcutaneous fat layer of the New Zealand white rabbits. The experiment aims to observe the cell types and adhesion to the gelatin layer through the environmental susceptibility of the stem cells.
- Results
- Perform a 10-minute observation (20 sec per image, a total of 30 images) for twice on the cells cultured with various concentrations of gelatin to monitor the adhesion rate of the cells to the gelatin layer. It is shown in the video that cells cultured in 0.1 % gelatin attach and spread within the first 10 minutes, while cells cultured in 10% gelatin remain suspended. It is known that the adhesion rate of the cells to gelatin decreases with the increase of the gelatin concentration.
- The adipose-derived stem cells were cultured with various concentrations of gelatin for 2 days until the cells had grown full of the plate to be examined for the cell amount under the same size of several areas and the differences in the cell morphology. It can be noticed from the images that the cell amounts of the groups cultured with 1%, 5%, and 10% gelatin are significantly greater than 0.1%. It is speculated that the growth rate of the cells increases in the gelatin layer, though the cell morphology are not much changed.
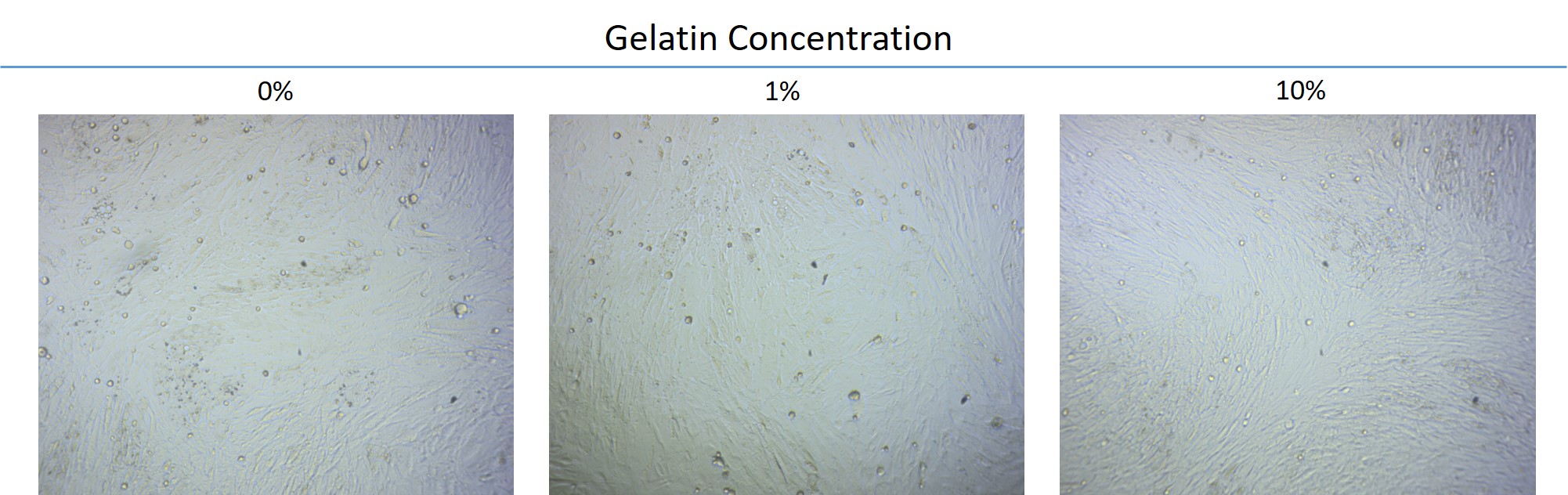
- The adipose-derived stem cells were cultured with various concentrations of gelatin and placed in an adipocyte differentiation medium for observation of 4 days. Whether the cells were cultured in gelatin or not, it is observed that the cells begin to produce oil drops and undergo differentiation into adipocytes. It can be concluded that gelatin does not affect the differentiation of the stem cells.
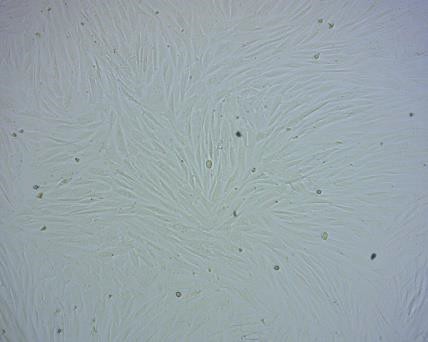
Photos of the original cellular condition and the same cell after experimental treatment
The original condition of the adipose-derived stem cells of rabbits:
The adipose-derived stem cells of rabbits cultured in gelatin:
Stem cell differentiation:
- Brief description of the follow-up research
According to the current observation, gelatin is nontoxic to cell growth, making it a good candidate for the biomaterial of the 3D bioscaffold. As a result, we have planned to use gelatin to prepare for the 3D bioscaffold in the follow-up project. Since the hardness and elasticity are two of the chief considerations regarding the bioscaffold, the follow-up research will be focused on the analyses of the mechanics and physical properties of the bioscaffold in order to identify the best concentration for regenerative studies in cartilage tissue engineering.


